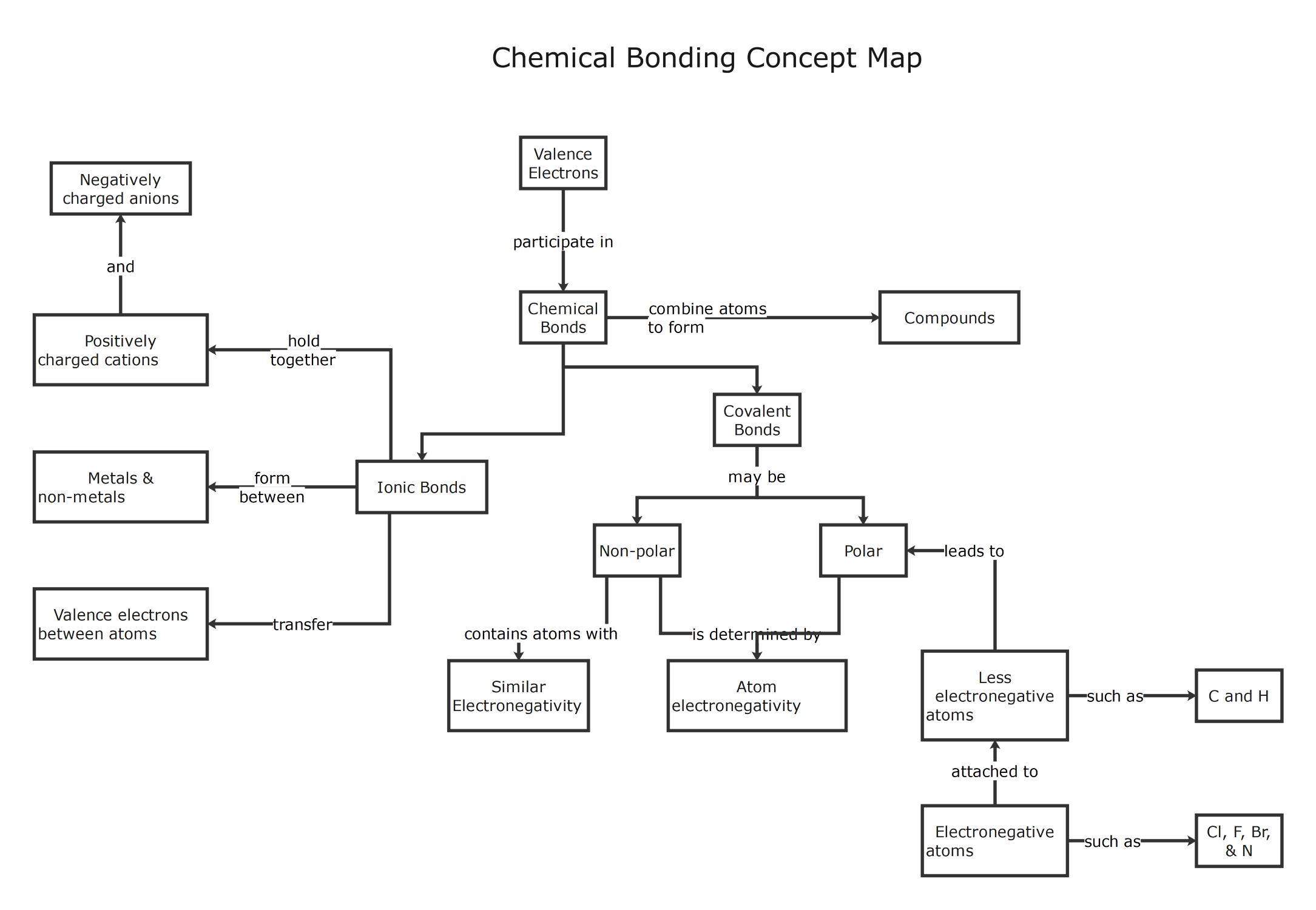
About this Chemical Bonding Concept Map template
This template provides a clear visual summary of the ways atoms interact through valence electrons. It helps students and educators map out the differences between ionic and covalent bonds while highlighting key characteristics like electronegativity and charge.
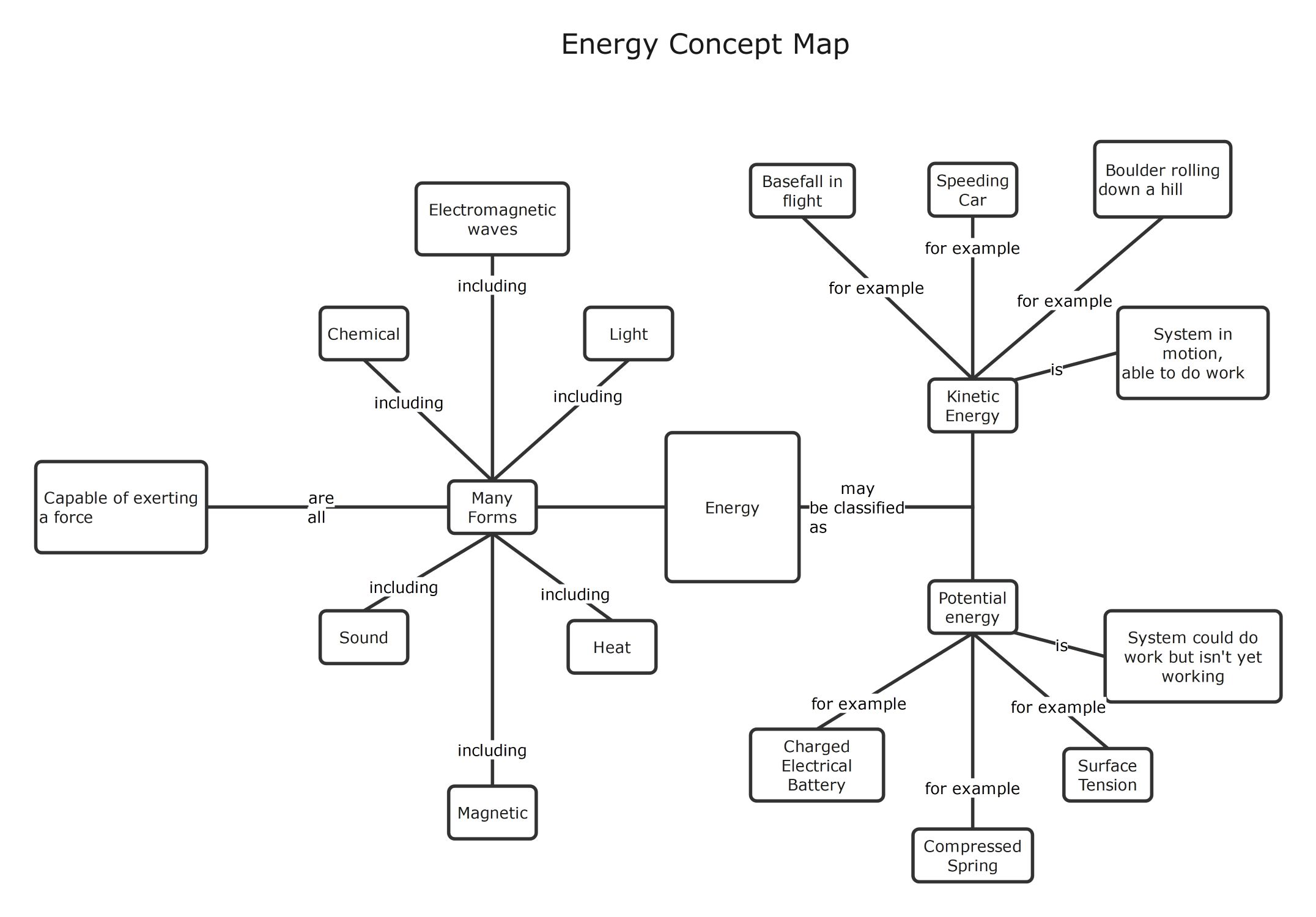
Chemical Bonds and Valence Electrons
Chemical bonds are formed when atoms interact through their outermost electrons to achieve stability. This process combines different atoms to create complex compounds which are the building blocks of all matter found in the universe.
- Valence Electrons
- Chemical Bonds
- Compounds
Ionic Bonds and Electron Transfer
Ionic bonds occur when electrons transfer between atoms, usually involving metals and non-metals. This attraction holds oppositely charged ions together to form stable structures that are often found in various common salts and minerals.
- Positively charged cations
- Negatively charged anions
- Metals and non-metals
- Valence electron transfer
Covalent Bonds and Electronegativity
Covalent bonds involve the sharing of electrons between atoms rather than a full transfer. The nature of this sharing depends heavily on electronegativity, which determines if the resulting bond is classified as polar or non-polar.
- Polar Covalent Bonds
- Non-polar Covalent Bonds
- Atom electronegativity
- Similar electronegativity
Polar Bond Characteristics and Examples
Polar bonds create unequal electron distribution when an electronegative atom pulls electrons toward itself. This section highlights common elements involved in these bonds, such as hydrogen and carbon paired with highly reactive halogen elements.
- Less electronegative atoms (C and H)
- Electronegative atoms (Cl, F, Br, and N)
- Unequal electron sharing
FAQs about this Template
-
What is the difference between ionic and covalent bonds?
Ionic bonds form when one atom gives up electrons to another, creating charged ions that attract each other. This typically happens between metals and non-metals. In contrast, covalent bonds involve atoms sharing electrons to stay together. This occurs most often between non-metal atoms. Understanding these two types of bonds is essential for predicting how different substances will behave.
-
How does electronegativity affect chemical bonding?
Electronegativity measures how strongly an atom attracts electrons. If two atoms have very different electronegativity values, they likely form an ionic bond. If the values are similar, they form a non-polar covalent bond. However, if there is a moderate difference, a polar covalent bond forms. This creates partial charges, which significantly influence the physical properties of the resulting molecule.
-
Why are valence electrons important in this concept map?
Valence electrons are the specific electrons located in the outermost shell of an atom. They are the primary players in chemical reactions and bonding. Because they are the most accessible, they determine how an atom will interact with its neighbors. The concept map highlights them because all bonding types, whether ionic or covalent, rely entirely on the behavior of these electrons.