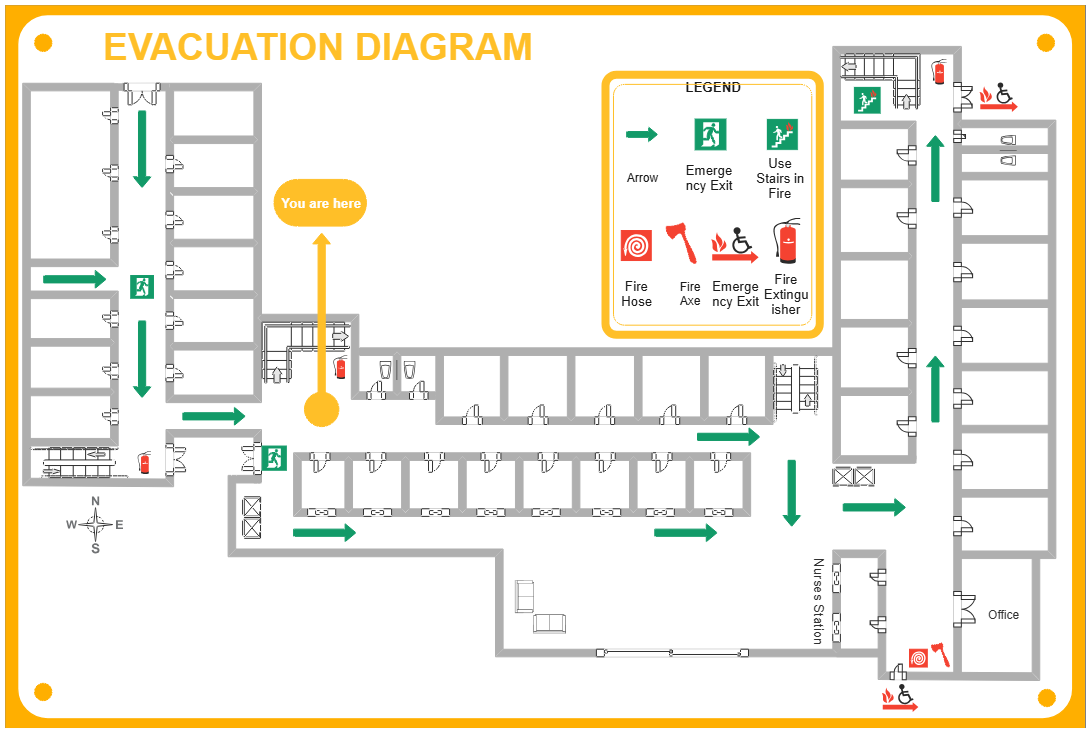
About this Atom Concept Map template
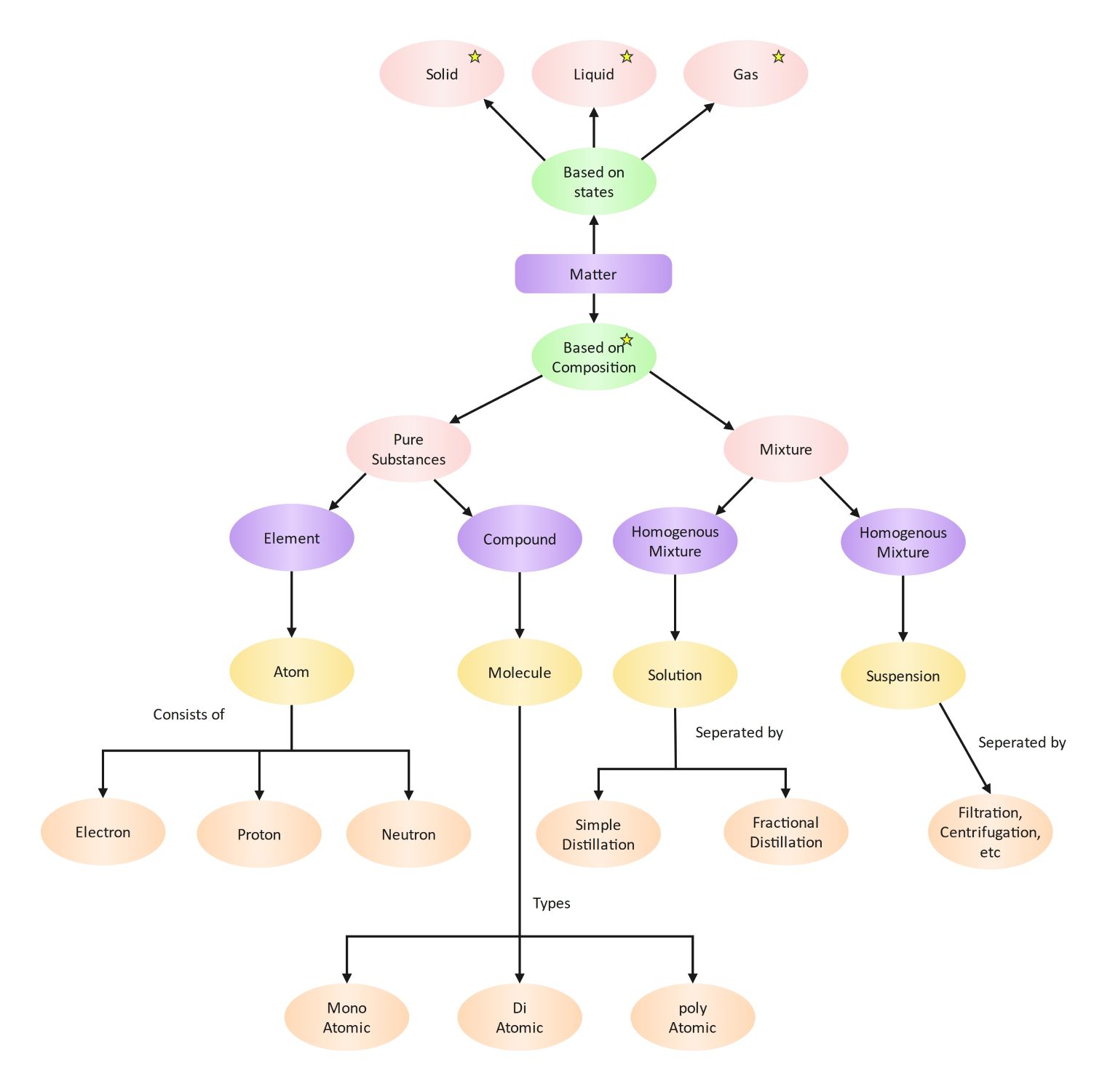
This Atom Concept Map template provides a visual framework for studying matter. It organizes the relationship between physical states and chemical compositions. Users can easily track how elements, compounds, and mixtures connect to form the world around us. It is an ideal resource for classrooms and personal study.
Classification by States
Matter is classified by its physical form into three main categories. These states describe how particles are arranged and how they move. Identifying these forms is the first step in understanding the physical properties of different substances.
- Solid
- Liquid
- Gas
Pure Substances and Atoms
Pure substances consist of only one type of particle with fixed properties. This section focuses on elements and their basic building blocks. It details the internal structure of atoms and the subatomic particles that give them their unique identities.
- Element
- Atom
- Electron
- Proton
- Neutron
Compounds and Molecular Forms
Compounds are formed when different elements chemically bond together. They exist as molecules, which are the smallest units of these substances. This part of the map categorizes molecules by the number of atoms they contain in their structure.
- Compound
- Molecule
- Mono Atomic
- Di Atomic
- Poly Atomic
Mixtures and Separation Techniques
Mixtures involve substances that are physically mixed but not chemically combined. This section explains how to classify these blends and the laboratory methods used to separate them. These techniques rely on the different physical properties of each component.
- Homogenous Mixture (Solution)
- Simple and Fractional Distillation
- Suspension (Heterogeneous Mixture)
- Filtration and Centrifugation
FAQs about this Template
-
Why is an atom concept map useful for learning chemistry?
An atom concept map is helpful because it organizes abstract ideas into a visual hierarchy. Chemistry involves many tiny particles and complex relationships that are hard to see. By mapping out how matter, atoms, and mixtures connect, students can better understand the big picture. This structure improves memory retention and makes it much easier to study for exams or laboratory assignments.
-
What is the difference between an element and a compound?
An element is a pure substance made of only one type of atom. It cannot be broken down into simpler substances by chemical means. A compound consists of two or more different elements that are chemically bonded together. While elements are listed on the periodic table, compounds have unique properties that are often different from the elements that make them up.
-
How does distillation separate parts of a solution?
Distillation separates components based on their different boiling points. When you heat a liquid mixture, the substance with the lower boiling point turns into vapor first. This vapor travels through a cooling tube where it condenses back into a liquid. By carefully controlling the temperature, scientists can isolate pure liquids from a complex solution. This method is very common in chemical labs.