About this Concept Map of Matter template
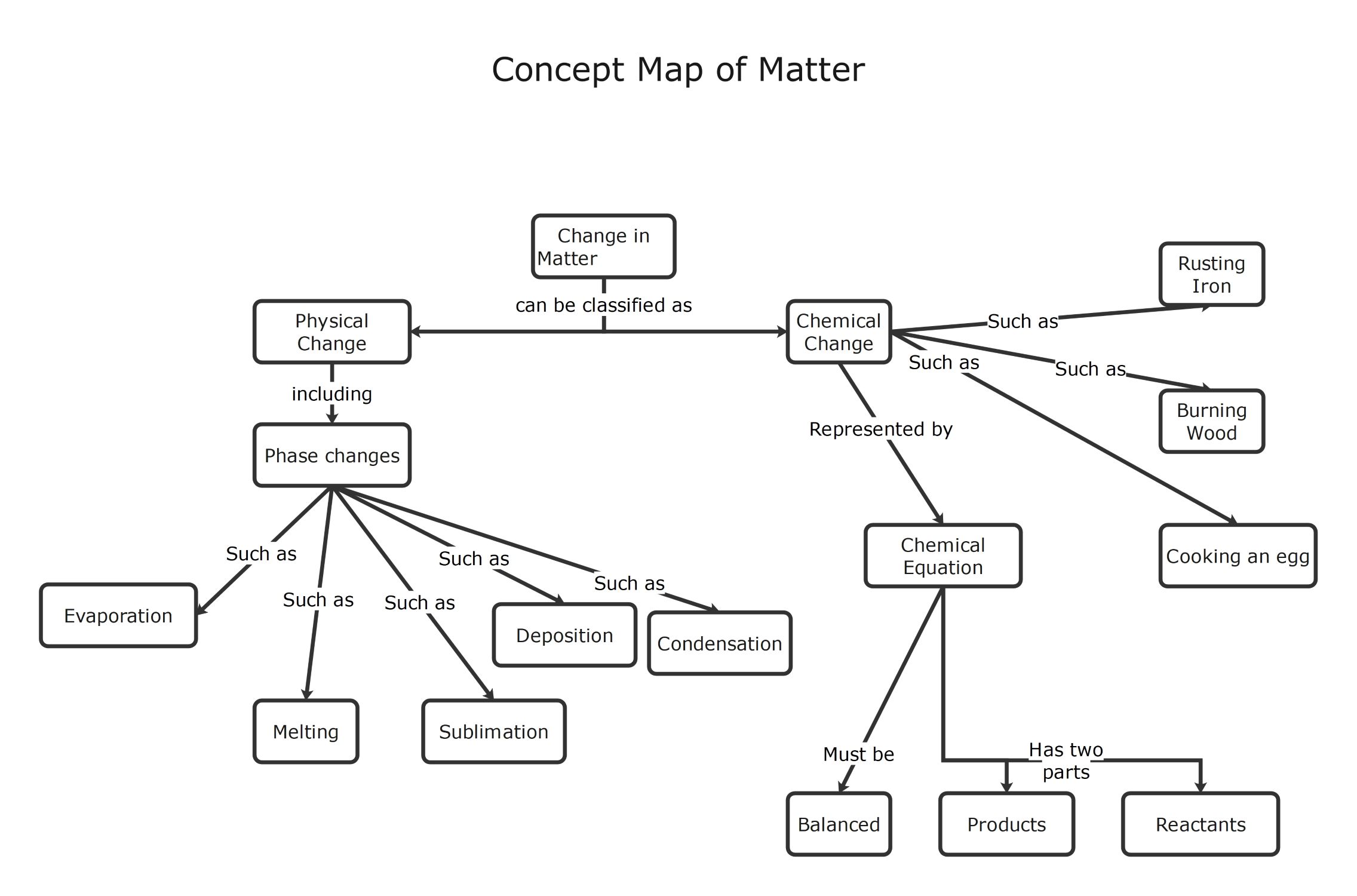
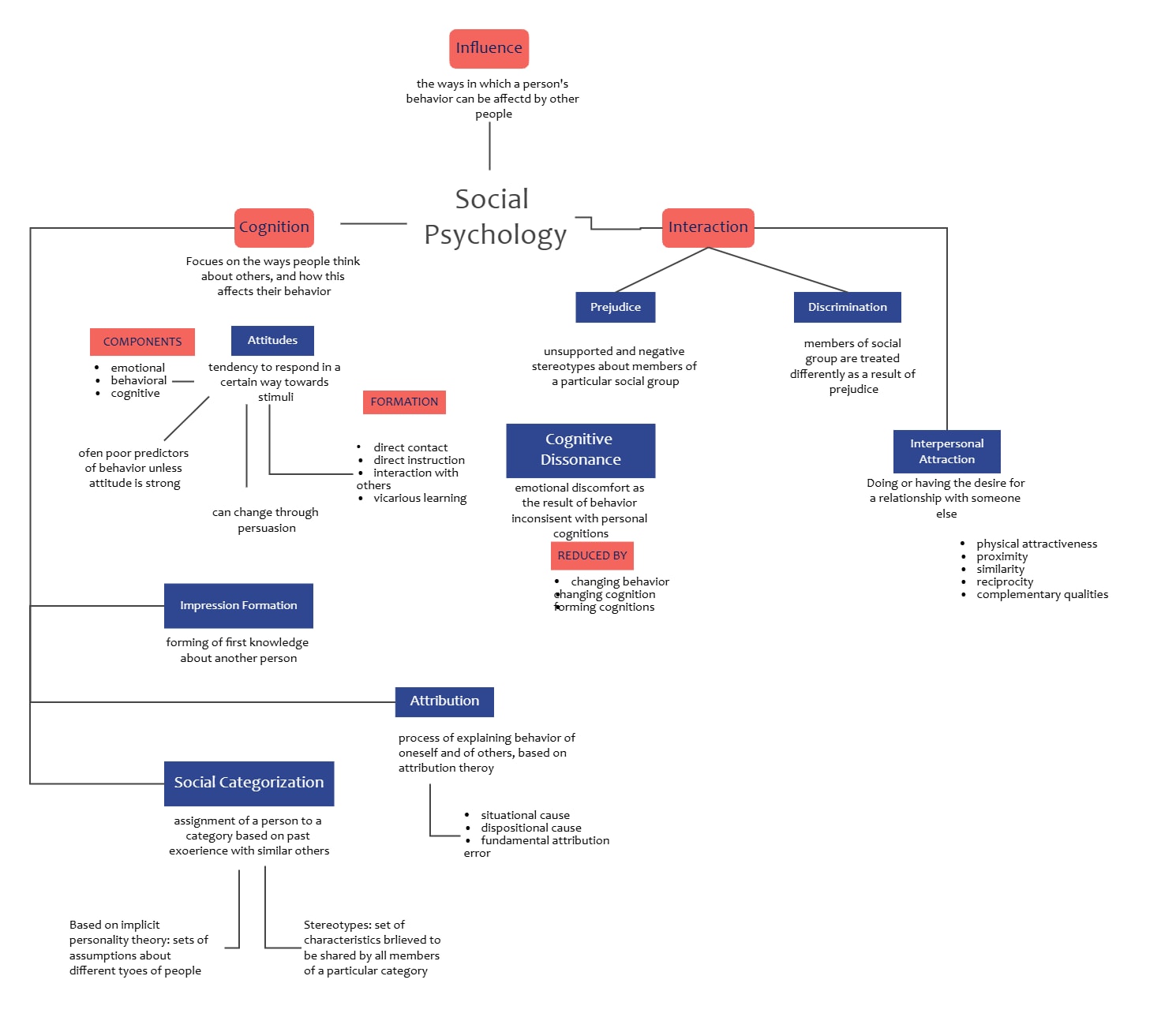
This template serves as a visual guide for understanding the transformations of matter. It clearly distinguishes between physical and chemical changes. Students can use it to learn about phase changes and chemical equations effectively.
Physical Change
Physical changes involve transformations that do not alter the chemical identity of a substance. They usually relate to state transitions where the material remains the same. These changes are often reversible and easily observed.
- Evaporation
- Melting
- Sublimation
- Deposition
- Condensation
Chemical Change
Chemical changes result in the formation of entirely new substances with different properties. These processes involve breaking or forming bonds. They are usually harder to reverse and produce specific observable signs like heat or color.
- Rusting Iron
- Burning Wood
- Cooking an egg
Chemical Equation
A chemical equation represents the symbolic notation of a chemical reaction. It shows how reactants turn into products while following the law of conservation of mass. Scientific accuracy requires these equations to be perfectly balanced.
- Balanced
- Products
- Reactants
FAQs about this Template
-
How do you identify a physical change in matter?
You can identify a physical change when the substance looks different but stays the same at a molecular level. Common examples include changes in shape, size, or state, such as ice melting into water. These changes do not produce new chemical products. Most of the time, you can reverse a physical change by simply altering the temperature or pressure of the environment.
-
What are the main signs of a chemical change?
Chemical changes often produce noticeable signs that a new substance has formed. You might see a change in color, the production of a gas through bubbling, or a distinct smell. In many cases, these reactions also release or absorb energy, resulting in temperature changes or light. Unlike physical changes, chemical reactions are typically permanent and cannot be undone by simple physical means.
-
Why is a balanced chemical equation important?
A balanced chemical equation is essential because it obeys the law of conservation of mass. This scientific law states that matter cannot be created or destroyed during a reaction. Therefore, the number of atoms for each element must be equal on both sides. This balance ensures that the equation accurately represents what happens in real life when reactants turn into products during a chemical change.